 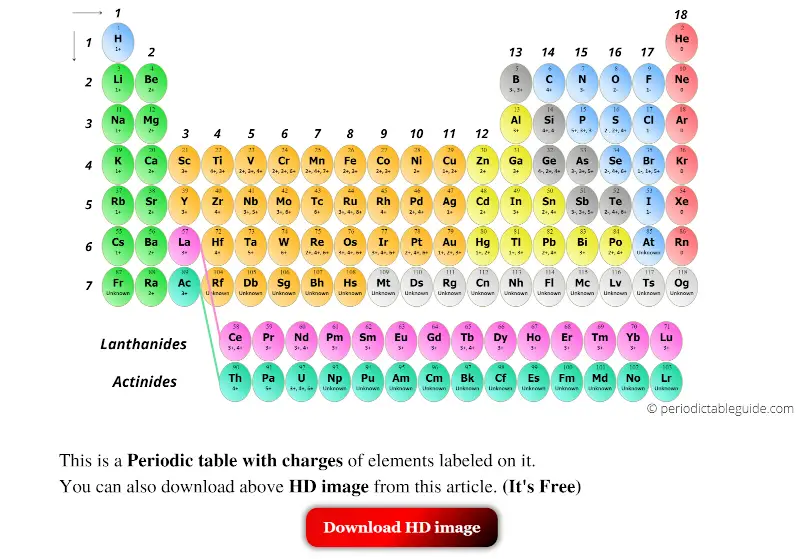
An element symbol without a charge written next to it is assumed to be the uncharged atom. We could write the chemical formula for this ionic compound as MgClCl MgClCl, but the convention is to use a numerical subscript when there is more than one ion of a given type MgCl2 MgCl 2. 
If the charge is a single positive or negative one, the number 1 is not written if the magnitude of the charge is greater than 1, then the number is written before the + or − sign. Now the positive and negative charges are balanced. The magnitude of the charge is listed as a right superscript next to the symbol of the element. The resulting sodium ion has the same electron configuration as neon (ls22s22p6) and has a + 1 charge, because there are 11 protons in the nucleus, but only 10. Answer a: Answer b: Answer c: Be aware that ionic compounds are empirical formulas and so must be written as the lowest ratio of the ions. Note the convention for indicating an ion. Write the chemical formula for an ionic compound composed of each pair of ions. Unfortunately, there is little understanding which two charges a metal atom may take, so it is best to just memorize the possible charges a particular element can have. 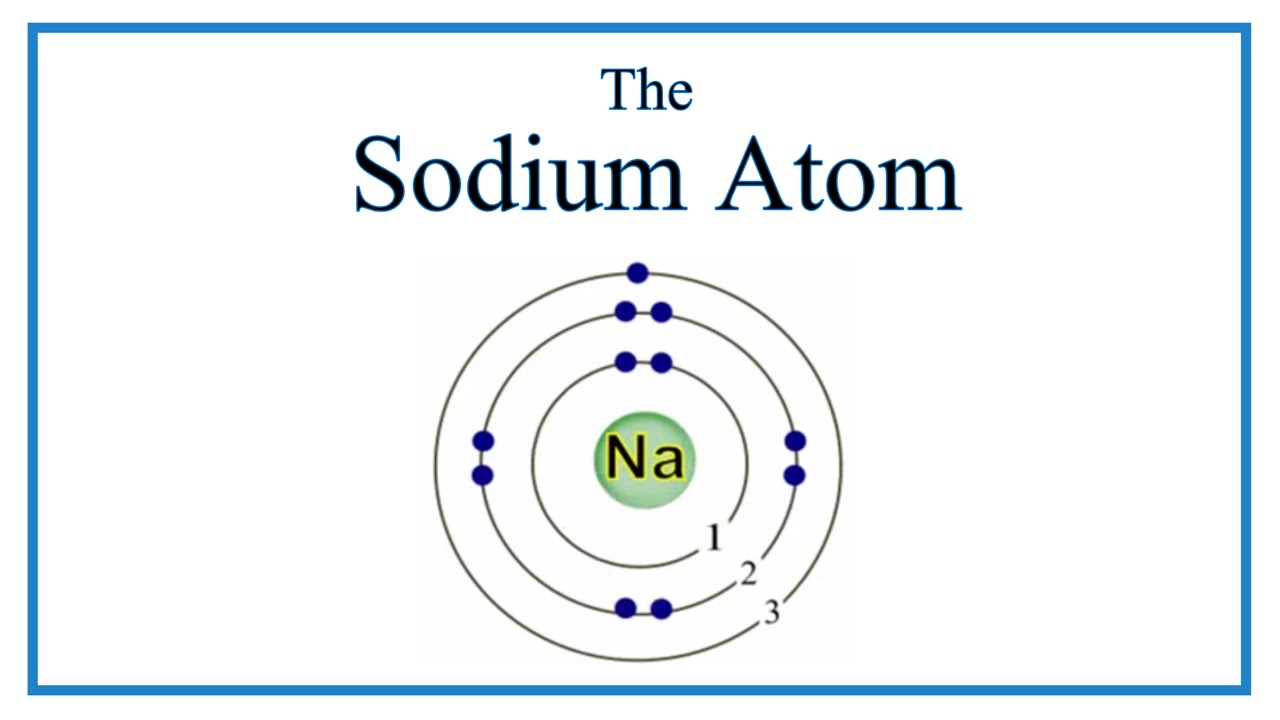
Cobalt (Co) is another element that can form more than one possible charged ion (2+ and 3+), while lead (Pb) can form 2+ or 4+ cations. For example, iron (Fe) atoms can form 2+ cations or 3+ cations. A few elements, all metals, can form more than one possible charge. The formula for sodium chloride, therefore, is NaCl since Na loses one electron and has a +1 charge and Cl gains one. Examination of the table in the link given shows that there are some exceptions to the previous point. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. A more complete table of ions and their charges can be found at Monotomic Ions of Various Charges. HTML5 simulations can run on iPads, Chromebooks, PC, Mac, and Linux systems. \) is helpful in determining the charge on a large number of our cations and anions it's hardly complete. As the sodium atom has equal numbers of protons and electrons, the charges cancel each other out, forming an overall neutral charge- this applies to all atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
